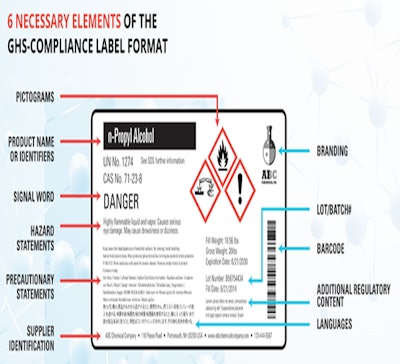
As the impending OSHA Hazard Communication Standard (HCS) deadline of June 15, 2015 approaches in the U.S., businesses are faced with the urgent need to define their strategy for the Globally Harmonized System of Classification and Labeling of Chemicals (GHS).
There are also a multitude of additional complexities confronting companies as they are asked to comply with more international regulations and customer requirements than ever before.
Here are seven key questions related to these challenges that organizations need to ask in moving forward with a GHS initiative.
Answers to these questions and challenges exist in the form of Enterprise Labeling Solutions, which offers an efficient and flexible way to meet the needs of GHS compliance, while also addressing a wide range of customer and regulatory requirements for doing business in the global supply chain.
Question #1: How Does My Company Plan to Deal with GHS?
This is an obvious question, since HCS and GHS regulations and standards have been in the making for a very long time, but now a hard and fast deadline is looming for accurately labeling products containing thousands of different hazardous chemicals. From raw materials to finished goods delivery, companies are facing more complexity in chemical labeling with risk of heavy penalties for non-compliance. Companies need to access multiple data systems and SDS applications, include 6 HCS elements, fill in all pictograms, fulfill regional regulatory requirements, include customer requirements, multiple languages, and more. How ready are you? Many companies have not spent enough time carefully reevaluating their overall labeling strategy to make it the best it can possibly be in a comprehensive, holistic way. Because equally importantly the question is, does your labeling process include the ability to deal with the complexity that extends well beyond the 2015 HCS and GHS requirements?
Solution #1: Enterprise Labeling Is Designed to Addresses Complex, Global Labeling Challenges, Including GHS, as Opposed to Only GHS.
Enterprise Labeling Solutions are designed to meet the rigors of GHS labeling while supporting large scale, global deployments with numerous products and packaging variations. This allows companies to leverage their source of truth for label data, design labels to meet the most complex requirements, enable data driven labeling to support different label sizes and variations, and empower companies to perform mission-critical, high volume printing. The best of these solutions stress the importance of labeling best practices and offer domain experience in managing the deployment of labeling solutions on a global basis. Enterprise Labeling solution providers also offer specific skills in implementing solutions in different industries and offer 24x7x365 customer support for an organization’s global operations.
Question #2: How Can We Implement One Effective, Scalable Labeling Strategy?
OSHA states, “Labels, as defined in The Hazardous Communications System (HCS), are an appropriate group of written, printed, or graphic informational elements concerning a hazardous chemical that are affixed to, printed on, or attached to the immediate container of a hazardous chemical, or to the outside packaging.” Every item on the factory or storeroom floor containing a hazardous chemical, and each shipment going to any location must be labeled within the precise guidelines for each destination. In other words, GHS compliance is only part of a robust labeling solution. The expanding global supply chain, customers’ change requests, multiple print locations, and frequently disparate labeling systems across organizations all point to a need for a more centralized yet flexible, scalable, and consistent approach to labeling.
Solution #2: Enterprise Labeling Ensures Consistency Throughout The Global Supply Chain to Meet All of Your Labeling Requirements.
Meeting labeling requirements might center on adherence to GHS standards, but there are other important considerations as well. Enterprise Labeling Solutions ensure consistency throughout the global supply chain, enable support for regional labeling considerations, provide supplier labeling capabilities, simplify adherence to customer specific labeling requirements, and minimize reliance on scarce IT resources by facilitating simple and rapid label changes. Instead of relying on disjointed and redundant labeling processes, Enterprise Labeling offers a scalable labeling solution to cover a multitude of labeling challenges, GHS-centric and beyond.
Question #3: How Can We Access The Labeling Data We Need From Our Regulatory and Transactional Systems?
With so many different types of data needed to create compliant labeling, organizations often find that multiple labeling systems with redundant data sources take root over time. SDS information, regulatory information, consistent branding, product identification, multiple languages, pictograms and more need to be included on labels. But with different labeling solutions scattered throughout the supply chain, inability to meet regulatory requirements, labeling errors, relabeling, and failure to address customer needs appropriately can lead to costly fines and—most importantly— loss of business. Getting the right data on all labels is critical.
Solution #3: Enterprise Labeling Offers the Ability to Integrate to Key Data Sources.
Enterprise Labeling Solutions are designed to drive your labeling directly from your source of truth for label data. Whether regulatory data is sourced from an SDS database or applications like SAP EHS Management, this approach provides the ability to integrate to your key data sources. At the same time, transactional data such as product information, lot and batch number, images or customer specific information can be sourced from content management or ERP systems like SAP or Oracle. This level of integration enables dynamic data-driven labeling, maximizes flexibility, and ensures accuracy without replication of data. Regulatory compliance, increased time and cost savings, plus quick response to market needs are the result.
Question #4: Is There a Way to Manage the Complexity of so Many Different Products, Shapes, Sizes and Requirements All at Once?
One of the major considerations in meeting GHS requirements is the number of possible permutations in labeling. Drums, pails, cans, cases, cartons, pallets, and even tiny vials must all be labeled with GHS compliant information. This means differently sized labels with different amounts of printing “real estate.” In addition to GHS-compliant attributes, proper information needs to be correctly represented on available label space along with additional regional regulatory data, customer and branding specifications, and language translations. In the chemical industry, the smallest packages and containers are especially challenging when it comes to creating the right label.
Solution #4: Enterprise Labeling Enables Sophisticated Field and Label Logic That Supports the Ability to Dynamically Determine Label Content.
Enterprise Labeling offers unmatched design capabilities that allow you to create any size or shape of label required, with out-of-box support for GHS pictograms. What’s more, some Enterprise Labeling Solutions allow for sophisticated field and label logic that supports the ability to dynamically determine label content based on available white space on the label. Rather than creating a myriad of labels for thousands of different chemical products and packaging variations, the power of a built-in rules engine enables label size, format and content to be determined based on transactional data.
Question #5: How Can Our Labels Accommodate All the Languages Needed for Regulatory Compliance Both in the US and Overseas?
Growth of the chemical industry has recently outpaced that of the U.S. economy with shipment values predicted to exceed $1 trillion in 2018, up from about $790 billion in 2013. Much of this growth will happen overseas with U.S. exports increasing to keep pace, so sophisticated language capabilities will continue to be centrally important to participants in the chemical industry supply chains. Making sure labels are compliant in any language is a fundamental consideration in today’s global marketplace.
Solution #5: Enterprise Labeling Offers Full Unicode Support Which Ensures that Any Language Can Appear Together on the Same Label.
Some Enterprise Labeling Solutions can provide unlimited support for languages that are an essential component of labeling in the global supply chain of the chemical industry. Full Unicode support ensures that any languages required for labeling, including languages that are read from right to left, can appear together on the same label. Advanced logic capabilities can calculate the real estate needed determine whether sufficient space exists to print a complete warning or text string on a label. This functionality greatly reduces the time and effort needed to produce accurate, compliant labels when delivering chemicals throughout a global supply chain.
Question #6: How Will We Manage Color Capabilities for GHS Label Printing?
What about the ability to support your need for color printing? For GHS compliance, whether by using pre-printed stock or color printers for pictograms and logos, you need a labeling solution that suits your color printing preferences. Consideration of the types of printers already installed, the cost of possible new printer acquisitions, and assessing the printing capabilities of distribution partners, resellers, and customers may also be a part of the decision making process. Managing countless variations of preprinted stock isn’t always easy, and the cost of retooling or coordinating between a wide array of printers throughout the supply chain can seem daunting as well.
Solution #6: Enterprise Labeling Offers Native Print Drivers to Support Color Printing.
For GHS labeling, pictograms require at least two colors, and Enterprise Labeling supports any approach to color printing that your organization prefers. For customers using pre-printed stock, sophisticated design capabilities can support the quality label printing results you need including the ability to leverage business logic to dynamically populate pre-printed pictograms based on transactional data. For those companies looking to embrace emerging color thermal and inkjet technologies, some Enterprise Labeling Solutions provide native print drivers for leading print technologies including Windows, PCL5c and cab support. Regardless of your approach, Enterprise Labeling offers the native support for thousands of different print devices to fulfill your GHS labeling requirements.
Question #7: How Can Our Labeling Strategy Include the Flexibility Required for Evolving Global Standards and Regulations?
Looking at the number of swiftly emerging chemical governmental regulations around the world, it is clear that labeling complexity is compounding everywhere. For example, the EU REACH & CLP of (2006/2008), Taiwan Existing Substance Inventory Available (2012), South Korea “K-REACH” (2015), Australia National Industrial Chemicals Notification and Assessment Scheme (NICNAS), and India’s draft of Hazardous Substances Rules—are just some of the many sets of regulations around the world in effect now or planned for implementation in the near future. Environmental concerns are dictating regulations with ongoing global pressure to improve safety standards in more countries.
Solution #7: Enterprise Labeling Leverages Data Driven Labeling and Business Logic to Respond to the Evolving Regulatory Landscape.
Change is the one thing you can count on in the global chemical supply chain, and Enterprise Labeling Solutions enable you to incorporate changing requirements with ease. Leveraging data driven labeling and a built-in rules engine, Enterprise Labeling provides support for new regulations and label specifications without requiring code or changes to a myriad of label templates. The end result is labeling agility and greater responsiveness to meet evolving labeling requirements without the assistance of expensive and scarce technical resources. Business logic even provides dynamic selection of labels and rapid support for regulatory variations through multi-channel supply chains, allowing for unprecedented levels of responsiveness.
If you’re interested in more detailed information about GHS and “The Challenges of Labeling for OSHA’s Revised Hazard Communication Standard,” please click on this link for a white paper from industry expert Daniel Levine, CHMM or to hear firsthand from Mr. Levine you can download a recording from our recent webinar “How to Manage Labeling Changes fro GHS Compliance.”



















